 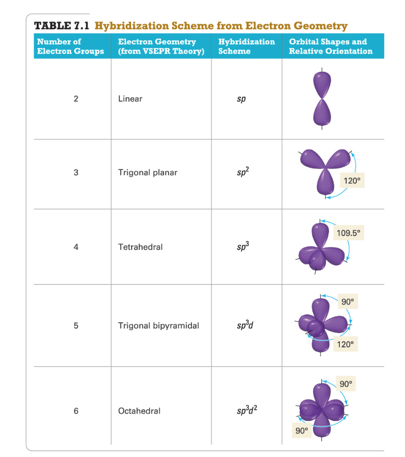
Lone pairs of electrons are found in nonbonding orbitals, i.e. Therefore, the carbon’s hybridization is sp. The carbon atom in CO2 has two double bonds, one with each atom of oxygen. An atom with two or more double bonds, or with a single triple bond, has a hybridization of sp. Does hybridization include double bonds?Īn atom with a single double bond has a hybridization of sp2. This intermixing usually results in the formation of hybrid orbitals having entirely different energies, shapes, etc. Hybridization in Chemistry is defined as the concept of mixing two atomic orbitals to give rise to a new type of hybridized orbitals. The numbers of atomic orbitals mixed together are always equal to the number of hybrid orbitals. The orbitals of almost the same energy level combine to form hybrid orbitals. The following are the rules related to hybridisation: Orbitals of only a central atom would undergo hybridisation. The two O−H bonds are formed from overlap of the sp3 hybrid orbitals from oxygen with the 1s atomic orbitals from the hydrogen atoms. Two of the four sp3 hybrid orbitals are used to form bonds to the two hydrogen atoms, and the other two sp3 hybrid orbitals hold the two lone pairs on oxygen. Double and triple bonds still count as being only bonded to one atom. An easy way to figure out what hybridization an atom has is to just count the number of atoms bonded to it and the number of lone pairs. The carbon is bonded to two other atoms, that means it needs two hybrid orbitals, aka sp. Which bonds are counted in hybridization? Quantum mechanics and atomic orbitals can give more sophisticated predictions when VSEPR is inadequate.24 How do you determine the hybridization of sigma and pi bonds? Why are lone pairs hybridized? VSEPR also predicts that group-2 halides such as will be linear when they are actually bent.For example, VSEPR predicts that and will have the same bond angles, but structural studies have shown the bonds in the two molecules are different by 12 degrees. First, the idealized bond angles do not always match the measured values.VSEPR is simple and useful but does not work for all chemical species.Rather, it is an algorithm that accurately predicts the structures of a large number of compounds. It does not explain or attempt to explain any observations or predictions. 
The overlap of the 1s orbital of the hydrogen atom and the 2p orbital of the fluorine atom makes the covalent bond in an HF molecule which is explained by the valence bond theory.One of its most important applications is the variation in the length and strength of chemical bonds in H 2 and F 2 molecules can be explained by the difference in their overlapping orbitals.The maximum overlap conditions which is given by the valence bond theory can explain the formation of covalent bonds in many compounds.Types of Hybrid Orbitals: Applications of Valence Bond Theory The pi bond is formed by the overlapping of atomic orbitals sideways, whereas the sigma bond is created by the overlapping of atomic orbitals along the axis of the nucleus. There are two types of covalent bonds based on the overlapping pattern: sigma bonds and pi bonds. 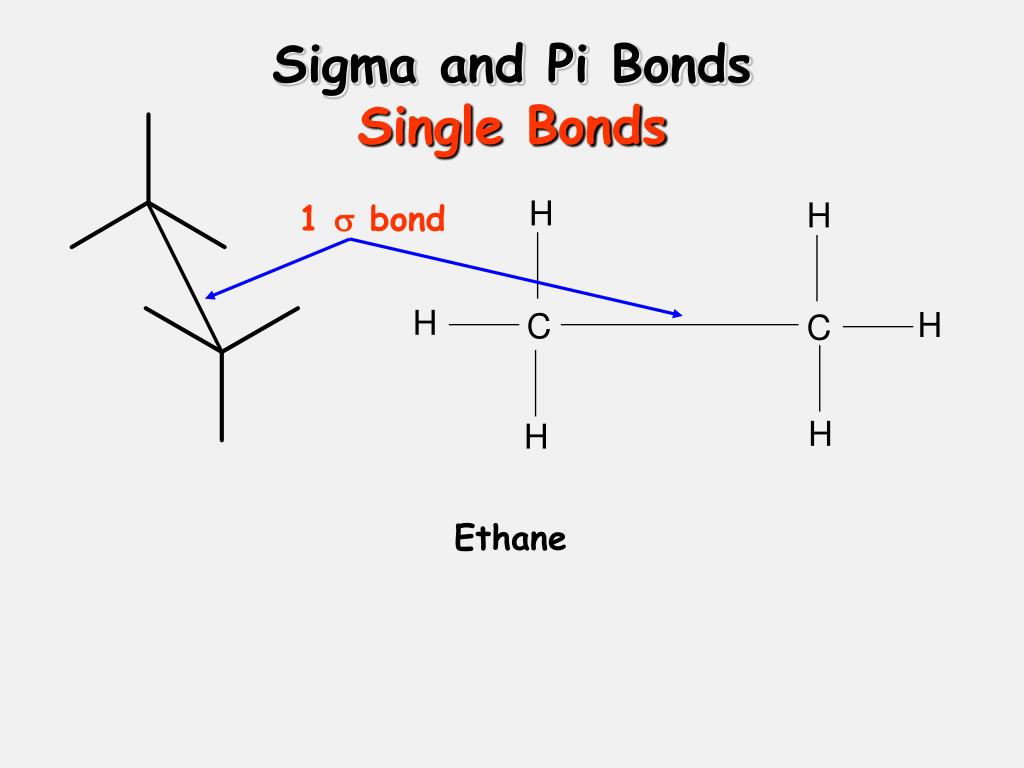
The region of overlapping atomic orbitals is also parallel to such a bond.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
